Cu0 Nanoparticles Deposited on Nanoporous Polymers: A Recyclable Heterogeneous Nanocatalyst for Ullmann Coupling of Aryl Halides with Amines in Water | Scientific Reports
Copper catalysed Ullmann type chemistry: from mechanistic aspects to modern development - Chemical Society Reviews (RSC Publishing)
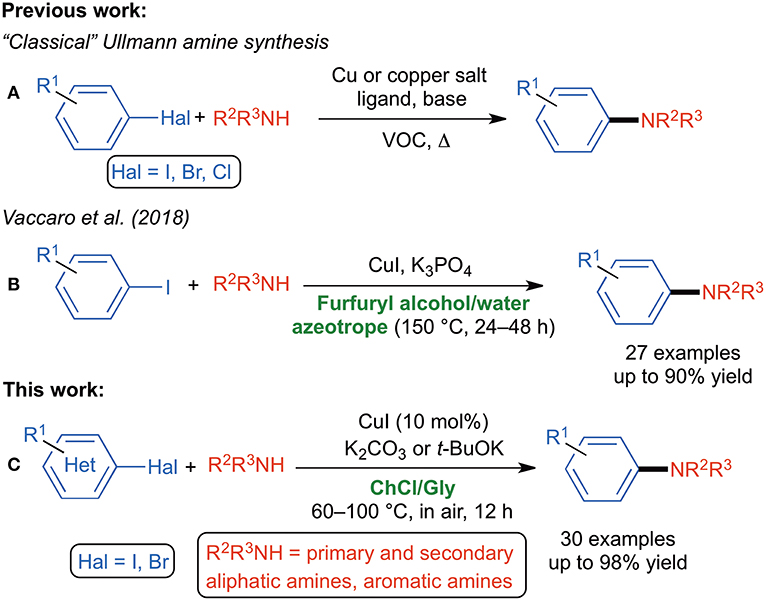
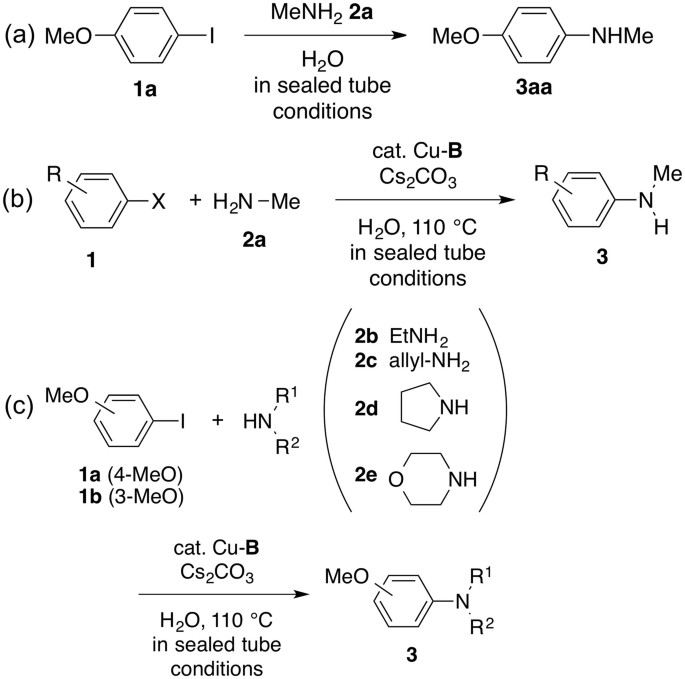
Frontiers | Reshaping Ullmann Amine Synthesis in Deep Eutectic Solvents: A Mild Approach for Cu-Catalyzed C–N Coupling Reactions With No Additional Ligands
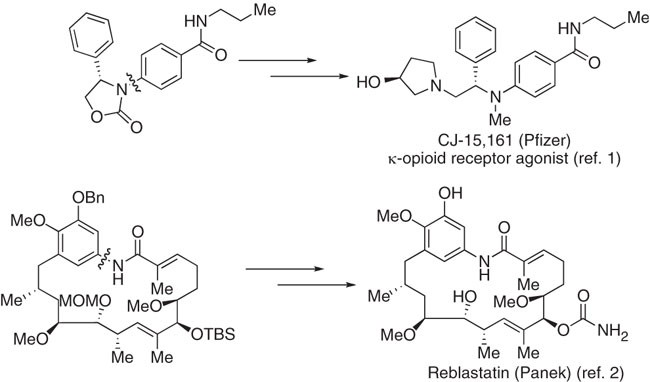
Cu-catalyzed Goldberg and Ullmann reactions of aryl halides using chelating N- and O-based ligands | Nature Protocols
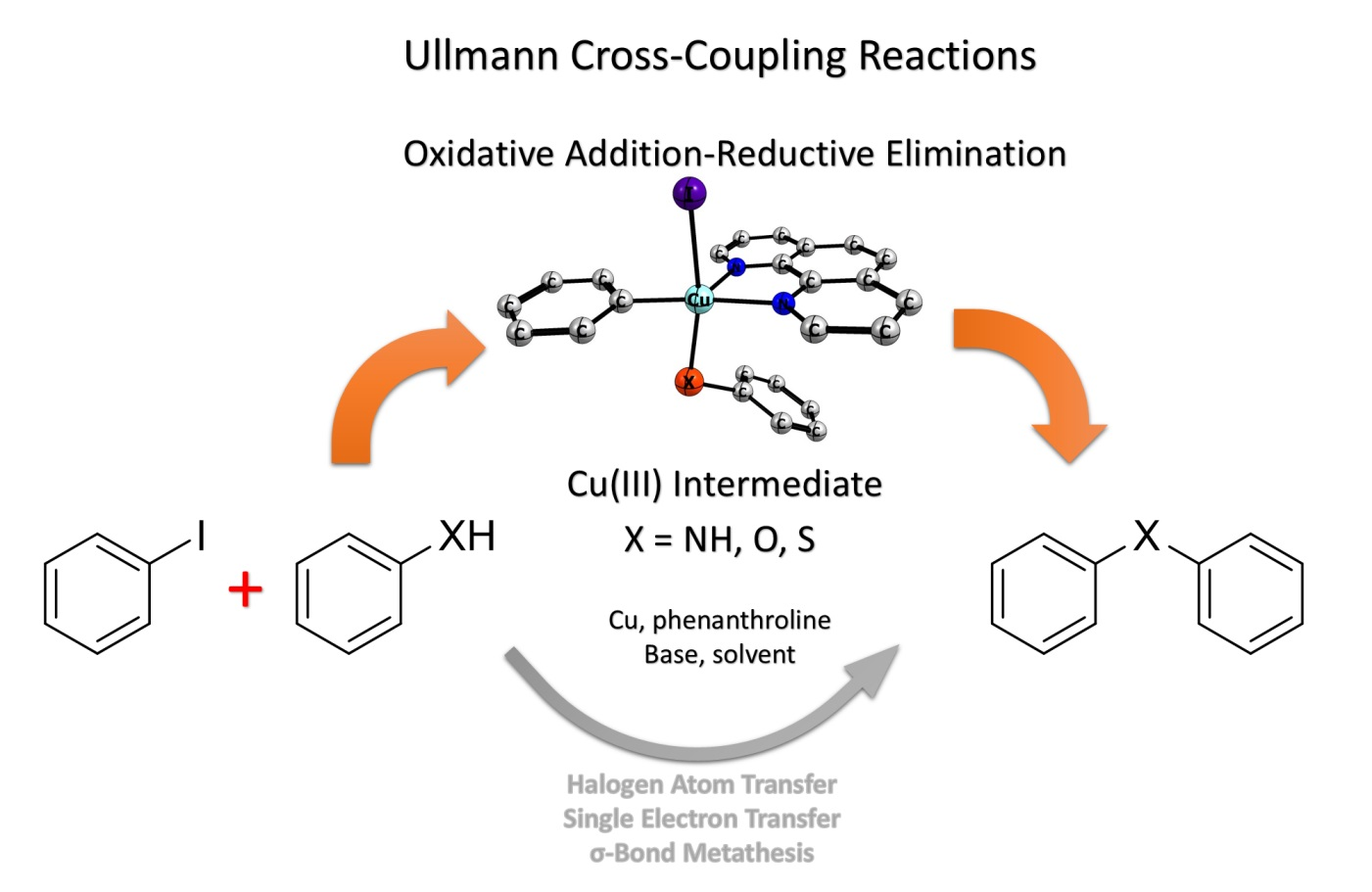
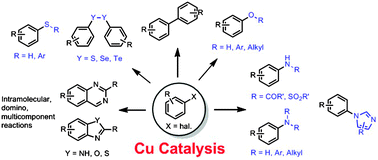
Catalysts | Free Full-Text | Understanding the Heteroatom Effect on the Ullmann Copper-Catalyzed Cross-Coupling of X-Arylation (X = NH, O, S) Mechanism

Preparation and characterization of isatin complexed with Cu supported on 4-(aminomethyl) benzoic acid-functionalized Fe3O4 nanoparticles as a novel magnetic catalyst for the Ullmann coupling reaction | SpringerLink

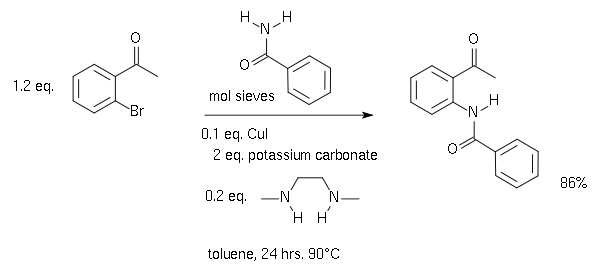

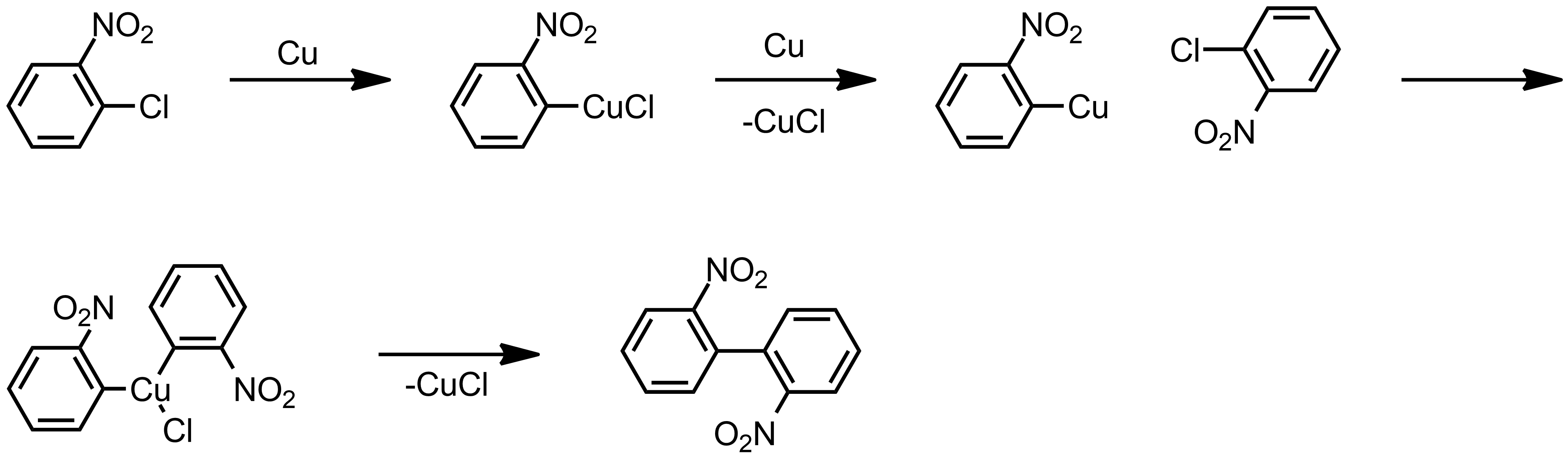
![97] Ullmann Aryl-Aryl Coupling 1901 – ChemInfoGraphic 97] Ullmann Aryl-Aryl Coupling 1901 – ChemInfoGraphic](https://cheminfographic.files.wordpress.com/2018/04/97_ullmann_aryl_coupling.jpg)



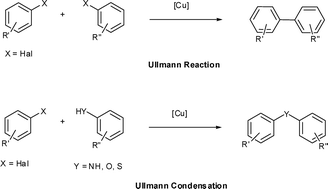





![PDF] The mechanism of the modified Ullmann reaction. | Semantic Scholar PDF] The mechanism of the modified Ullmann reaction. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/5708eada114a1981b8c1ed8ad8dd20d120d5725e/8-Figure3-1.png)